- Use the term half-life in simple calculations, including the use of information in tables or decay curves.
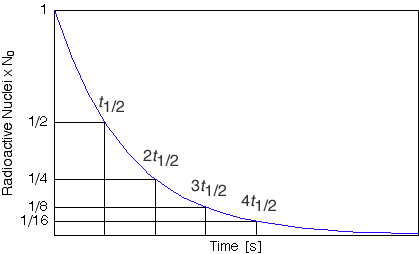
The half-life of a radioactive substance is the time it takes for parent nuclei in a sample to halve. In other words, half-life of a radioactive substance is the time it takes for the count rate (on a Geiger-Muller counter) from the original substance to fall to half its initial level.
If we start with 1000 unstable nuclei and 10% disintegrate every hour, we would expect 100 nuclei to decay in the first hour leaving 900. Another 10% (900*10%=90) will decay in the next hour leaving 810 and so on. If we were to draw a table to record this decay, it would look like this:

From the table above we can calculate the half-life of this particular radioactive substance to be somewhere between 6 to 7 hours (half of 1000 is 500 and it falls to 500 nuclei in the seventh hour).
In one half-life the number of nuclei decreases by half. In the second half-life it decreases by a further half and so on.
Notes submitted by Lintha
Click here to go to the next topic.
Click here to go back to the previous topic.
Click here to go back to the Science menu.