- State the meaning of radioactive decay.
When a nucleus is unstable, it will disintegrate (break up). This shoots out tiny particles and a burst of energy. These materials are radioactive, so the breaking up of the nucleus is called radioactive decay. Each unstable nucleus changes into an isotope of a different element after radioactive decay.
- Use equations (involving words or symbols) to represent changes in the composition of the nucleus when particles are emitted.
When a nucleus emits an alpha (α) particle, it loses 2 protons and 2 neutrons. This is denoted by the symbol 42α. 4 denotes the total number of nucleons (protons and neutrons) emitted and 2 denotes the number of protons emitted. Example:
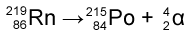
The radon isotope with 219 nucleons and 86 protons has given out an alpha particle of 4 nucleons and 2 protons to become an isotope of Polonium with a nucleon number of 215 and proton number of 84. (You can find out what element’s isotope it becomes by looking at which element has a proton number that is two times lesser than the original element, in the periodic table).
When a nucleus emits a beta (β) particle, an electron are emitted and a neutron in the nucleus changes into a proton. This is denoted by the symbol 0-1β. 0 denotes that there is no emission of any nucleon. -1 denotes the electron emitted (as well as the proton gained instead of emitted). Example:

The Carbon isotope with 14 nucleons and 6 protons has given out a beta particle of 1 electron to become an isotope of Nitrogen with 14 nucleons and 7 protons. (You can find out the new element formed by looking at the periodic table to find out which element has a proton number that is one greater than the original element)
When a nucleus emits gamma (γ) radiation, the number of protons and neutrons remains the same; no changes occur.
Notes submitted by Lintha
Click here to go to the next topic.
Click here to go back to the previous topic.
Click here to go back to the Science menu.