- Describe the formation of poly(ethene) as an example of addition polymerisation of monomer units.
The process by which monomers join together to form polymers (macromolecules) is called polymerisation. Polymers are often formed through addition reactions. Where there is only one polymer formed in a reaction, it is called addition polymerisation. The formation of poly(ethene) is an example
The product formed when ethene monomers join together in a long chain is called poly(ethene). When the ethene molecules react, the double bonds between the two carbon molecules break. Note that carbon molecules have a valency of four – so now that the double bonds are broken, carbon needs to form a fourth bond to become stable, so they react with the carbon in other ethene monomers. When thousands of these ethene molecules join, poly(ethene) is formed.
-
- Draw the structure of poly(ethene).

In condensation polymerisation, the monomers react together to form a polymer and another small molecule such as water or hydrogen chloride. Condensation polymerisation usually involves two different monomers, each with a different functional group (-OH, -COOH), or one monomer which has both the different functional groups.
-
- Describe the formation of a simple condensation polymer exemplified by nylon, the structure of nylon being represented as:
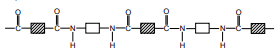
- Describe the formation of a simple condensation polymer exemplified by nylon, the structure of nylon being represented as:
Nylon is called a polyamide. Polyamides are formed through the condensation polymerisation of a carboxylic acid (which has a -COOH functional group) and an amine (a compound with an NH2 functional group). The diagram below demonstrates the formation of a single amide from an amine and a carboxylic acid – not the formation of a polyamide. The carboxylic acid reacts with the amine to form an amide (functional group CONH):
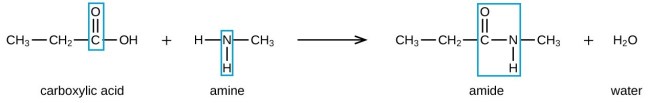
By the same mechanism as above, a polyamide is formed (as shown in the diagram below). The difference between the above and below reaction is that each monomer has two functional groups, one on each end so they can form a chain that extends from both sides. These monomers have the word di- in front of them (because they each have two of the same functional group) and you can represent them as:

So the full structure of nylon will look like this:

Notes submitted by Lintha and edited by Sarah.
Click here to go to the next topic.
Click here to go to the previous topic.
Click here to go back to the Science menu.
These notes are very helpful and Thanks a lot :)) !!! Just a few grammar errors here and there but still your notes indeed stand out..
LikeLike
Thank you! We’re happy they help 🙂
Thank you for pointing out that mistakes are present, too – I’ve just gone through and edited it so it should hopefully be fine now. If you spot any more mistakes, please don’t hesitate to tell us 🙂
Good luck with your exams!
LikeLiked by 1 person